آخرین روش های استفاده از استخراج کننده مس
استفاده از استخراج کننده مس در سال های اخیر, با پیشرفت تکنولوژی و افزایش قوانین زیست محیطی, روش های استفاده از استخراج کننده های مس به طور مداوم تکامل یافته و بهبود یافته است. به طور گسترده ای در ذوب فلز استفاده می شود, فرآوری سنگ معدن و حفاظت از محیط زیست.
در زیر برخی از جدیدترین روشهای استفاده از استخراجکنندههای مس آورده شده است:
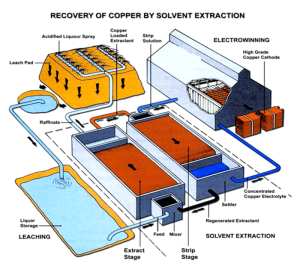
1. بهینه سازی روش استخراج با حلال
استخراج کننده های مس مدرن معمولا از روش های استخراج با حلال استفاده می کنند, که شامل استراتژی های بهینه سازی زیر می باشد:
- استخراج انتخابی: استخراجکنندههای انتخابی جدید برای افزایش گزینش پذیری یونهای مس ایجاد شدهاند, در نتیجه استخراج همزمان سایر فلزات مانند سرب و روی کاهش می یابد.
- استخراج مرحله ای: با تنظیم غلظت ماده استخراج کننده و مقدار pH, استخراج مرحله ای از محلول را می توان برای بهبود نرخ بازیابی مس انجام داد.
2. رویکردهای شیمی سبز
با افزایش آگاهی از حفاظت از محیط زیست, کاربردهای شیمی سبز در استخراج مس به تدریج در حال افزایش است:
- استخراج بیولوژیکی: استفاده از میکروارگانیسم ها یا گیاهان خاص برای استخراج مس از سنگ معدن یک روش سازگار با محیط زیست و پایدار است.. برخی از میکروارگانیسم ها می توانند به طور موثر یون های مس را جذب کرده و در فرآیندهای بعدی مس را فیلتر یا رسوب دهند..
- استخراج بدون حلال: توسعه روش های استخراج که به حلال های آلی نیاز ندارند, با استفاده از مواد جامد (مانند رزین های تبادل یونی) استخراج مستقیم مس از محلول ها.
3. تحقیق و توسعه عصاره گیری های جدید
محققان به طور مداوم انواع جدیدی از استخراج کننده ها را برای افزایش کارایی و انتخاب پذیری استخراج می کنند:
- نانو مواد: استفاده از نانوذرات به عنوان استخراج کننده به طور قابل توجهی سطح و سرعت واکنش را افزایش می دهد, بهبود راندمان استخراج یون مس.
- مواد کاربردی: با معرفی گروه های عاملی آلی به مواد معدنی, استخراج کننده های جدید با عملکردهای خاص را می توان تهیه کرد.
4. بهینه سازی شرایط فرآیند
بهینه سازی شرایط فرآیند در طول استخراج مس نیز برای بهبود اثربخشی بسیار مهم است:
- کنترل دما و زمان واکنش: با تنظیم دمای عملیاتی و زمان واکنش, سینتیک استخراج مس را می توان بهینه کرد, افزایش راندمان استخراج.
- تنظیم سرعت هم زدن: تنظیم صحیح سرعت همزدن می تواند راندمان تماس بین استخراج کننده و یون مس را بهبود بخشد, بنابراین سرعت استخراج را تسریع می کند.
5. نظارت و کنترل آنلاین
فناوریهای نظارت آنلاین مدرن، فرآیند استخراج مس را کارآمدتر و هوشمندتر میکند:
- تجزیه و تحلیل زمان واقعی: استفاده از سنسورهای پیشرفته و ابزارهای تحلیلی برای نظارت بر غلظت مس و سایر پارامترها در زمان واقعی, تنظیم پویا مقدار استخراج کننده و شرایط فرآیند.
نتیجه گیری در مورد استفاده از استخراج کننده مس
جدیدترین روش های استفاده از استخراج کننده مس به سمت راندمان بالاتر پیش می رود, دوستی با محیط زیست, و هوش. این روش های جدید نه تنها نرخ بازیافت مس را بهبود می بخشد، بلکه اثرات زیست محیطی را نیز کاهش می دهد, ارائه راهکارهای جدید برای استفاده پایدار از منابع مس. با تحقیقات مداوم و پیشرفت های تکنولوژیکی, حوزه استخراج مس در آینده شاهد نوآوری ها و پیشرفت های بیشتری خواهد بود.
استخراج کننده های فلزی ما به شرح زیر است, در صورت نیاز به هر نوع, لطفا از ما بپرسید:
- P204 (D2EHPA یا HDEHP) این برای اولین مرحله برای حذف ناخالصی برای سنگ معدن نیکل لاتریت استفاده می شود.
- DY319 استخراجکننده هماستخراج نیکل کبالت با راندمان بالا برای بازیافت باتری, می تواند نیکل و کبالت را با هم از الکترولیت باتری لیتیوم خارج کند.
- DY272 استخراج کننده نیکل کبالت, می تواند کبالت را از محلول نیکل کبالت خارج کند, سپس نیکل خالص را ترک کنید.
- DY988N/DY973N/DY902/DY5640 معرف استخراج با حلال مس.
- P507 استخراج کننده فلزات غیر آهنی برای مس, فلز روی, کبالت نیکل, کادمیوم, طلا-نقره, فلزات گروه پلاتین, خاک های کمیاب و غیره.
- DY377 استخراج کننده کارآمد جداسازی نیکل و الماس.
- DY366 استخراج کننده اسکاندیم.
- DY316 استخراج کننده لیتیوم.
- DY-SS-01 استخراج کننده برای آهن, کلسیم, کادمیوم و اسکاندیم در مقادیر مختلف pH.

