A New Process for Efficient Recovery of Valuable Metals from Calcium Magnesium Containing Solutions
As the demand for lithium-ion batteries continues to expand, the demand for lithium cobalt oxide, lithium nickel oxide, and lithium nickel cobalt manganese oxide as cathode materials for lithium-ion batteries has also greatly increased. At present, abundant mineral resources are almost exhausted, while the number of waste lithium-ion batteries is increasing rapidly. If waste lithium batteries are not recycled, it will result in a significant waste of valuable metal resources. Therefore, achieving the recycling and reuse of nickel cobalt manganese lithium metal is a necessary means of properly treating waste lithium-ion batteries.
At present, there are many methods for recovering valuable metals from the leaching solution of waste lithium-ion batteries, such as chemical precipitation method, hydrometallurgy method, etc. However, when multiple metals coexist, using chemical precipitation method to recover nickel, cobalt, and manganese in the cathode material of waste ternary lithium-ion batteries will cause a small amount of calcium and magnesium co deposition, resulting in a decrease in the purity of the recovered products. Hydrometallurgy has the characteristics of low energy consumption, low pollution, high resource utilization rate, and high product purity, which has been continuously studied and developed by many researchers.
At present, in the method of recovering valuable metals from waste lithium batteries using hydrometallurgical processes, P204 extractant is generally used to first recover Mn and a small amount of Fe, Zn, Cu, Al. After reverse extraction, a manganese sulfate solution is obtained. After further purification, qualified Mn products are obtained. Then, P507 extractant is used to extract Co from the raffinate, and CoSO4 solution is obtained after reverse extraction crystallization to obtain CoSO4 · 7H2O product. Finally, P507 is used to extract Ni from the raffinate, After reverse extraction crystallization, NiSO4 · 6H2O product was obtained. However, some leaching processes introduce some calcium and magnesium, which makes it impossible for P507 to directly prepare qualified NiSO4 · 6H2O products. Therefore, C272 needs to be further used to purify nickel sulfate. This process involves multiple extractions, which increases the cost of separately recovering nickel, cobalt, manganese, and lithium elements, and the separation process is also relatively long.
The order of pH 50 for P204 extraction of metal ions is Ca2+>Mn2+>Mg2+, Co2+, Ni2+. Therefore, P204 extraction cannot simultaneously achieve the separation of Mn2+, Co2+, Ni2+from Ca2+, Mg2+.
The DY319 extractant produced by our company has the following characteristics:
1. It is possible to extract Ni2+before extracting Mg2+and Ca2+, which has a good separation effect on Ni2+and Mg2+, thus achieving the goal of one-step purification of NiSO4;
2. Capable of synchronously extracting Co2+and Ni2+, with high extraction efficiency and good separation effect with Mg2+, Ca2+, Li+;
3. This product is easy to extract and can achieve efficient recovery of Co2+and Ni2+;
4. In actual use, there is less loss, low water solubility, reduced operating costs, and improved economic benefits;
5. In the early stage, impurities such as Fe3+, Zn2+, Cu2+, Al2+, Cr2+, Cd2+, Pb2+can be removed completely, and synchronous extraction of Co2+, Ni2+, and Mn2+can also be achieved.
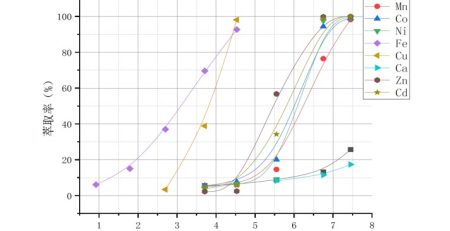
The extraction sequence of metal ions pH50 by DY319 is Cu2+>Zn2+>Ni2+≈ Co2+≈ Mn2+>Mg2+≈ Ca2+, which can achieve the separation of Ni2+, Co2+, Mn2+from Mg2+, Ca2+.

If you are interested in our experiment, please contact us for detailed experimental data.
We special to focus on R&D metal extraction reagents, our major products as below:
- DZ988N/DZ973N/DZ902 copper solvent extraction reagent.
- DZ272 Nickel, cobalt, manganese, and magnesium separation extractant.
- DY319 high efficiency nickel cobalt co extraction extractant.
- DY377 efficient nickel and diamond separation extractant.
- DY366 new advanced nickel cobalt extractant.
- P204 (D2EHPA or HDEHP) extractant.
- DY301, DY302 for nuclear spent fuel recovery.
- Other extraction reagents for Vanadium extractant, Lithium extractant, Ferro extractant and rare earth extractant.