DY-SS-01 extractant for iron, calcium, cadmium and scandium is an amide acid extractant, also can be used for the extraction of nickel, cobalt and rare earth metals.
The following is a detailed introduction to DY-SS-01 extractant:
|
DY-SS-01 specification |
|
| Item |
Specification |
| Appearance |
Yellow oily transparent liquid |
| Concentration (%) |
≥50 |
| Density (25℃) g/ml |
0.910-0.930 |
| Dynamic Viscosity (25℃) CP |
<60 |
| Dynamic Viscosity (50℃) CP |
<20 |
| Flash Point (℃) |
>80 |
DY-SS-01 extractant for iron, calcium, cadmium and scandium is a specially designed synthetic organic compound belonging to the carboxylic acid or phosphonic acid chelating extractant class. Its molecular structure contains functional groups that can form stable chelates with metal ions, thereby achieving the selective “capture” of specific metal ions from aqueous solutions.
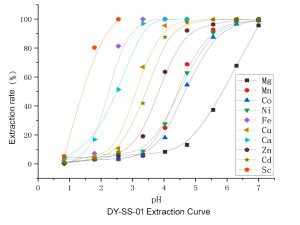
Its core performance characteristics are as follows:
1. Wide pH adaptability and selective extraction: Different metal ions have different optimal pH values for forming chelates with DY-SS-01. Therefore, by precisely controlling the pH value of the aqueous phase, selective separation of multiple metals can be achieved.
• Iron (Fe³⁺) extraction: Iron can usually be efficiently extracted even in strongly acidic environments (pH < 2). This makes it very suitable for preferentially removing iron in sulfuric acid systems (such as leaching solutions from hydrometallurgical zinc refining), creating conditions for subsequent processes.
• Scandium Extraction (Sc³⁺): Scandium, a rare and valuable rare earth element, can be efficiently and selectively extracted by the DY-SS-01 extractor within a weakly acidic to neutral pH range (pH 2.5 – 4.5). This is a crucial step in recovering scandium from complex solutions such as red mud, titanium dioxide waste acid, and tungsten slag.
• Cadmium (Cd²⁺) and Calcium (Ca²⁺): These divalent metal ions are typically extracted effectively at higher pH values (neutral to weakly alkaline). This makes it possible to separate or concentrate cadmium from solutions containing calcium and magnesium.
In simple terms, the extraction sequence can be summarized as follows: first extract iron at a low pH, then increase the pH to extract scandium, and then further increase the pH to extract cadmium and calcium. This characteristic makes it possible to separate multiple valuable metals from complex solutions using a “pH gradient extraction” process. Of course, in most cases, these metals do not coexist.
2. Easy Back-Extraction: An excellent extractant must not only “hold” the metal firmly but also “release” it easily. The chelates formed by DY-SS-01 with metals exhibit moderate stability, allowing for easy “recapture” of metal ions using stronger acids, thus achieving back-extraction.
• For metal-loaded organic phases, back-extraction with higher concentrations of inorganic acids (such as hydrochloric acid or sulfuric acid) is sufficient to efficiently back-extract the metal ions into a new aqueous phase.
• For example, iron-loaded organic phases can be back-extracted using 6M HCl; scandium-loaded organic phases can be back-extracted using 2-4M HCl or H₂SO₄.
• This characteristic allows for the recycling of the organic phase, significantly reducing operating costs and yielding a high-purity metal concentrate, facilitating subsequent electrolysis or precipitation treatment.
3. High Selectivity: Compared to traditional extractants such as P₂O₄, DY-SS-01 exhibits better selectivity for certain specific ions (e.g., scandium relative to other rare earth elements, and iron relative to other impurities), contributing to the acquisition of higher purity products.
4. Excellent Physicochemical Properties:
• Typically dissolved in diluents such as kerosene and sulfonated kerosene, it has a large density difference with the aqueous phase, facilitating phase separation.
• Relatively stable chemical properties, not easily degraded or emulsified, ensuring long-term stable operation in continuous industrial production.
Main Application Areas
Based on the above characteristics, DY-SS-01 extractant for iron, calcium, cadmium and scandium is mainly used for:
1. Recovery and purification of the rare metal scandium: This is one of its most important applications. It efficiently and selectively extracts and purifies scandium oxide from secondary resources such as titanium dioxide waste acid, tungsten slag, red mud, and chlorinated flue dust.
2. Iron removal in hydrometallurgy: Effectively separates and removes iron from solutions during the hydrometallurgical processes of cobalt, nickel, and zinc, purifying the electrolyte.
3. Heavy metal wastewater treatment: Used to treat industrial wastewater containing heavy metals such as cadmium, achieving heavy metal recovery and ensuring compliant water discharge.
4. Rare Earth Grouping and Separation: In rare earth separation processes, it can be used to group or separate certain specific rare earth elements.
Our metal extractants as below, if you need any kind, please ask us:
- P204 (D2EHPA or HDEHP) This is used for first step to remove impurity for laterite nickel ore.
- DY319 high efficiency nickel cobalt co-extraction extractant for battery recycle, can take out nickel and cobalt together from Lithium battery electrolyte.
- DY272 Nickel cobalt separation extractant, it can take cobalt out from nickel cobalt solution, then leave pure nickel.
- DY988N/DY973N/DY902/DY5640 copper solvent extraction reagent.
- P507 non-ferrous metal extractant for copper, zinc, cobalt-nickel, cadmium, gold-silver, platinum group metals, rare earths and so on.
- DY377 efficient nickel and diamond separation extractant.
- DY366 Scandium extractant.
- DY316 Lithium extractant.







